PSMA Therapy (Lutetium-177) – Radioligand Therapy for Advanced Prostate Cancer
PSMA therapy is an advanced treatment option for men with advanced or metastatic prostate cancer. It is typically used when the disease has progressed despite surgery, hormone therapy, or chemotherapy.
The therapy is based on a key characteristic of many prostate cancer cells: the so-called prostate-specific membrane antigen (PSMA). This protein is often present in increased amounts on the surface of tumor cells. The treatment specifically targets this feature.
A radiolabeled therapeutic agent - most commonly Lutetium-177 (¹⁷⁷Lu) - is administered via the bloodstream. It binds selectively to PSMA-positive cancer cells. The emitted radiation acts directly at the tumor site and has only a very short range in the surrounding tissue. This allows metastases in bones or lymph nodes to be specifically targeted.
A prerequisite for treatment is sufficient PSMA expression, which is confirmed by PSMA PET/CT imaging. Only if the tumor cells express this target is the therapy considered appropriate.
Today, PSMA therapy is included in international clinical guidelines - such as those of the European Association of Urology (EAU) - as a treatment option in advanced stages of the disease. The VISION study (NEJM, 2021) demonstrated that, in selected patients, median overall survival could be significantly prolonged.
It is important to understand: In the metastatic setting, PSMA therapy is not curative. The goal is to slow disease progression and to extend survival while maintaining quality of life. Whether it is suitable in an individual case is carefully evaluated based on the specific clinical indication.
What Is PSMA Radioligand Therapy?
PSMA therapy is a form of so-called radioligand therapy (RLT). In this approach, a targeted molecule is linked to a radioactive substance. This molecule binds to the prostate-specific membrane antigen (PSMA), which is highly expressed on many tumor cells in advanced prostate cancer.
As a first step, PSMA PET/CT imaging is used to determine whether the cancer cells express sufficient PSMA. This examination shows where active tumor cells are located in the body. If this biological requirement is met, radioligand therapy can be applied in a targeted manner.
The radiolabeled therapeutic agent - most commonly Lutetium-177 - is administered via a vein. It circulates throughout the bloodstream and binds to PSMA-positive tumor cells. At these sites, it releases radiation that damages the cancer cells.
This close integration of diagnostics and therapy is known as theranostics. In other words, the target structure is first visualized and then specifically used for treatment.
Whether this therapy is medically appropriate is carefully evaluated based on the individual indication for PSMA therapy.
How Does Lutetium-177 Work on Tumor Cells?
In PSMA therapy, the radioactive isotope Lutetium-177 (¹⁷⁷Lu) is most commonly used. It is a so-called beta emitter, meaning it releases radiation that travels only a very short distance within tissue.
Once the therapeutic agent binds to a PSMA-positive tumor cell, it releases radiation directly at that site. This radiation damages the genetic material (DNA) of the cancer cell. As a result, the cell can no longer divide or dies.
The range of this radiation is on average about 0.3 millimeters. This means that primarily the affected tumor cell and its immediate surroundings are targeted, while more distant healthy tissue is largely spared. This principle distinguishes PSMA therapy from external beam radiation, where radiation passes through healthy tissue from the outside.
Because the therapeutic agent circulates via the bloodstream, multiple metastases can be targeted simultaneously—for example, in bones or lymph nodes. This systemic approach is particularly important in metastatic prostate cancer.
A prerequisite for this targeted effect is the prior confirmation of sufficient PSMA expression using PSMA PET/CT imaging.
Which Patients Are Candidates for PSMA Therapy?
PSMA therapy is a treatment option for men with advanced or metastatic prostate cancer. It is typically used when the disease continues to progress despite established therapies.
This usually applies to situations in which:
- Metastases are present, for example in bones or lymph nodes
- Hormone therapy is no longer sufficiently effective
- And, in many cases, additional systemic treatments have already been administered
At this stage of the disease, the primary focus is on controlling disease progression. PSMA therapy is not a first-line treatment in early-stage disease, but rather part of a structured treatment sequence.
A key prerequisite is that the tumor cells express sufficient PSMA. This is verified using PSMA PET/CT imaging. Only if this biological condition is met can the therapy be expected to work effectively.
The specific medical criteria and detailed decision-making process are outlined on the indication for PSMA therapy page. There, it is explained which factors are taken into account in each individual case.
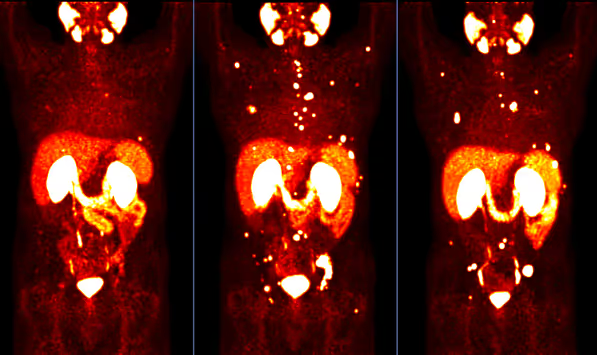
How Effective Is PSMA Therapy?
The effectiveness of PSMA therapy has been evaluated in several international studies. The most important and largest study to date is the VISION trial, published in the New England Journal of Medicine in 2021 (Sartor et al., NEJM 2021).
In this phase III study, men with metastatic, castration-resistant prostate cancer received Lutetium-177 PSMA either alone or in addition to established standard therapy.
The results showed:
- Median overall survival was 15.3 months in the PSMA therapy group, compared to 11.3 months with standard therapy alone
- Radiographic progression-free survival was also significantly prolonged
- Disease progression could be delayed more frequently
These data demonstrate that PSMA therapy can provide a measurable clinical benefit in appropriately selected patients. It is therefore not an experimental treatment, but a scientifically well-validated option in advanced disease.
A realistic perspective remains essential:
The therapy is not curative, but aims to control the disease and extend survival while maintaining quality of life. Not all patients respond equally to treatment, which is why a structured evaluation of the indication for PSMA therapy is crucial.
Additional studies - such as the TheraP trial - further support the effectiveness of Lutetium-177 PSMA therapy, including at earlier stages of the disease course.
Advantages of PSMA Therapy
In advanced prostate cancer, PSMA therapy offers several important advantages. These are primarily based on its targeted mechanism of action.
Targeted Treatment at the Molecular Level
The therapy utilizes a specific target structure on tumor cells—the PSMA. This allows radiation to be delivered directly to the cancer cells. Unlike conventional chemotherapy, which affects all rapidly dividing cells, PSMA therapy specifically targets cells that express PSMA.
Systemic Effect in Metastatic Disease
Because the therapeutic agent circulates via the bloodstream, it can reach metastatic lesions throughout the body—for example, in the skeleton or lymph nodes. This is particularly relevant in advanced disease, where localized treatment alone is often not sufficient.
Scientifically Proven Effectiveness
The effectiveness of PSMA therapy has been evaluated in large clinical trials. The VISION study demonstrated a significant improvement in median overall survival in appropriately selected patients. The therapy is therefore based on robust scientific evidence.
Personalized Treatment Approach
Before treatment, PSMA PET/CT imaging is used to determine whether tumor cells express sufficient PSMA. This approach increases the likelihood that the therapy will be effective. A detailed medical evaluation is carried out as part of the indication for PSMA therapy.
Despite these advantages, careful individual assessment remains essential. The therapy is not suitable for every patient and does not replace curative treatment options in early-stage disease.

Limitations of PSMA Therapy
As effective as PSMA therapy can be in advanced stages, it also has clear medical limitations.
Not a Curative Treatment in Metastatic Disease
In metastatic prostate cancer, PSMA therapy is not a curative treatment. The goal is to slow disease progression and extend survival. Complete remission is generally not achievable at this stage of the disease.
Not Every Tumor Is Suitable
For the therapy to be effective, tumor cells must express sufficient PSMA. If expression is low or the target structure is absent, treatment efficacy may be limited. This is assessed in advance using PSMA PET/CT imaging.
Part of a Treatment Sequence
PSMA therapy is typically not used in early, localized stages of the disease. In earlier phases, curative approaches such as surgery or radiation therapy are the primary focus. Radioligand therapy is generally part of a later stage in the treatment pathway.
Individual Risk–Benefit Assessment
Like any systemic therapy, PSMA therapy may cause side effects. A structured evaluation of potential benefits and risks is therefore essential. A detailed overview of possible side effects can be found on the Side Effects of PSMA Therapy page.
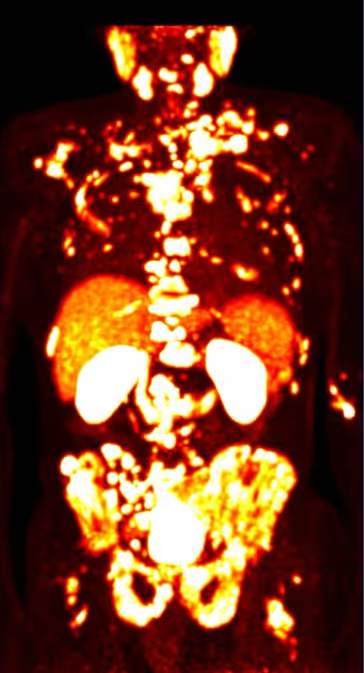
How Is PSMA Therapy Performed?
PSMA therapy is carried out in multiple treatment cycles. The goal is to achieve a therapeutic effect while regularly monitoring tolerability.
Administration via Infusion
The radiolabeled therapeutic agent - most commonly Lutetium-177 PSMA - is administered intravenously. The infusion itself typically takes only a few minutes and is followed by a short period of medical observation.
Treatment in Multiple Cycles
The therapy is usually performed in several sessions at intervals of a few weeks. Between the individual cycles, blood values are monitored and the response to treatment is assessed.
Monitoring Treatment Response
Disease progression and treatment response are monitored through laboratory tests (such as PSA levels) as well as imaging studies. PSMA PET/CT is often used to objectively evaluate the response.
The exact number of treatment cycles and the organizational details depend on the individual clinical situation. A detailed description of the individual steps can be found on the PSMA Therapy Process page.
Safety and Side Effects
Overall, PSMA therapy is considered to be well tolerated. However, it is an effective systemic treatment that can cause side effects.
Common side effects include:
- Fatigue or reduced physical resilience
- Temporary changes in blood counts
- Dry mouth
Because the therapeutic agent circulates through the bloodstream, the bone marrow may also be temporarily affected. For this reason, regular laboratory monitoring is carried out between treatment cycles.
The severity of side effects varies from person to person. It depends, among other factors, on overall health status, tumor burden, and any prior treatments.
A detailed and medically structured overview of potential risks can be found on the Side Effects of PSMA Therapy page.
Costs and Coverage of PSMA Therapy
PSMA therapy is a specialized nuclear medicine procedure. The costs consist of several components, including careful preparation and patient selection, the radiopharmaceutical (Lutetium-177 PSMA), close clinical monitoring, medical services, as well as accompanying follow-up tests and examinations.
In Austria, cost coverage is often determined on a case-by-case basis. A key factor is whether there is a clear, guideline-based indication for PSMA therapy. An up-to-date PSMA PET/CT scan is typically part of the medical evaluation.
For privately insured patients, coverage depends on the individual insurance plan. For self-paying or international patients, a transparent cost estimate is usually provided in advance.
It is important to note that cost-related decisions are always based on a thorough medical assessment.
PSMA Therapy at Theranosticum Vienna
The administration of PSMA therapy requires extensive experience in nuclear medicine radioligand therapy, as well as a structured medical evaluation in advance. What matters is not only the technical execution of the infusion, but also the careful integration of findings into the individual course of the disease.
At Theranosticum Vienna, treatment follows the principle of theranostics:
First, a PSMA PET/CT scan is performed to determine whether the tumor cells sufficiently express PSMA. Based on these findings, a decision is made as to whether the therapy is appropriate.
Before each treatment, a structured assessment of the indication for PSMA therapy is carried out. This includes consideration of prior treatments, current findings, and the patient’s overall health status.
The therapy itself is performed according to standardized protocols. Between treatment cycles, regular laboratory monitoring and clinical evaluations are conducted. A detailed description of the organizational process can be found under Procedure of PSMA Therapy.
The goal is a clear, evidence-based treatment plan with precise medical interpretation.
Frequently Asked Questions about PSMA Therapy (FAQ)
Is PSMA Therapy Curative?
No. In metastatic prostate cancer, PSMA therapy is not curative. The goal is to slow disease progression and extend overall survival.
How Long Does PSMA Therapy Work?
The effect varies between individuals. Clinical studies have shown that disease progression can often be significantly delayed. The duration of response depends on the course of the disease and prior treatments.
How Many Treatment Cycles Are Typical?
PSMA therapy is usually administered in multiple cycles spaced several weeks apart. The exact number depends on treatment response and tolerability.
When Can Treatment Response Be Expected?
An initial response may be reflected in declining PSA levels. However, a reliable assessment is based on imaging - most commonly PSMA PET/CT.
Is PSMA Therapy an Alternative to Chemotherapy?
PSMA therapy is based on a fundamentally different mechanism of action than chemotherapy. The most appropriate treatment depends on the individual stage of disease and prior treatment history.
We take time for what matters - your care
If you have questions or would like to schedule an appointment, we are here for you.
In a personal consultation, we discuss your concerns in a calm setting and outline the next steps together.
Our team will get back to you promptly and support you competently and reliably - from the first contact through to your therapy appointment.

We're here for you
Do you have questions or would you like to schedule an appointment?
We take the time to understand your concerns and will get back to you promptly.
Contact Details
Do you have questions about PSMA therapy? Contact our team for further information or to schedule an appointment.
Phone
Adress
Heiligenstädter Str. 55/65
1190 Vienna - Austria



