Indication for PSMA Therapy
Whether PSMA therapy is appropriate depends on several medical prerequisites. The decision is not made automatically, but is based on a structured evaluation of the disease course, previous treatments, and current findings.
In general, the indication applies to men with advanced or metastatic prostate cancer in whom established treatment options are no longer sufficiently effective. A further requirement is confirmation of sufficient PSMA expression by means of PSMA PET/CT.
The assessment of suitability is guided by international guidelines, particularly the recommendations of the European Association of Urology (EAU), as well as current scientific evidence. Medical criteria are systematically evaluated to realistically determine whether the therapy is likely to provide a meaningful benefit in the individual case.
This structured indication assessment not only serves to identify suitable patients, but also to prevent treatment that is unlikely to offer sufficient benefit. Our transparent evaluation provides clarity and helps patients make treatment decisions based on a well-founded medical basis.
Basic Requirement: Advanced or Metastatic Prostate Cancer
A key prerequisite for the indication for PSMA therapy is the presence of advanced or metastatic prostate cancer.
In early stages of the disease, where the tumor is confined to the prostate, curative treatment options such as surgery or radiation therapy are usually the primary focus. In this setting, PSMA therapy is generally not considered appropriate.
Suitability for PSMA therapy is typically considered when:
- Metastases are present, for example in bones or lymph nodes
- The disease progresses under hormone therapy
- Progression is observed despite additional systemic treatments
- Or upon patient request
At this stage of the disease, the focus is on controlling a systemic condition. PSMA therapy is part of a structured treatment sequence and is used only when other established therapies are no longer sufficiently effective or are not desired.
Assessment is always carried out on an individual basis and takes the entire clinical picture into account. The goal is to use the therapy only when a realistic medical benefit can be expected. This decision is made together with you.
Confirmation of PSMA Expression by PSMA PET/CT
A central prerequisite for eligibility for PSMA therapy is confirmation that the tumor cells express sufficient PSMA. Without this target structure, the treatment is not meaningful.
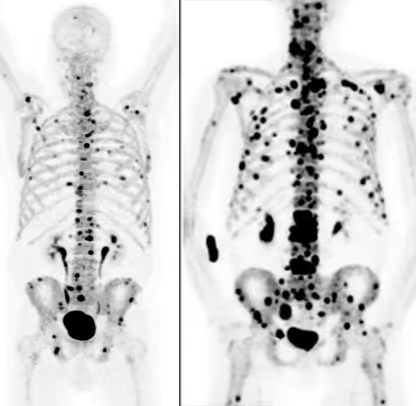
This confirmation is obtained through PSMA PET/CT. This specialized nuclear medicine examination shows whether and to what extent tumor lesions take up PSMA. At the same time, it reveals where active metastases are located in the body.
Only if imaging demonstrates clear PSMA uptake in the tumor lesions is the therapy considered biologically appropriate. If this expression is absent or only minimal, the effectiveness of the treatment is limited or not expected.
PSMA PET/CT is therefore not an additional component, but a fundamental requirement of the indication assessment. It ensures that the therapy is used only when a realistic effect can be expected.
This structured evaluation helps to avoid unnecessary burden and ensures that treatment is applied specifically where it is medically appropriate.
Previous Treatments and Course of Therapy
Suitability for PSMA therapy is always assessed in the context of the prior treatment history. It is generally not a first-line therapy, but is considered when established treatment options are no longer sufficiently effective.
This typically applies to patients with metastatic prostate cancer in whom:
- Hormone therapy has lost its effectiveness
- Modern anti-hormonal medications have been used
- and often when chemotherapy has already been administered
In this situation, the disease course is often referred to as castration-resistant. PSMA therapy is only appropriate if the disease continues to progress despite these treatments.
For the evaluation of indication, this means that individual values alone are not decisive—the overall clinical picture is key. PSA trends, current imaging, and clinical condition are assessed together.
This structured approach ensures that the therapy is recommended only when it can be meaningfully integrated into the existing treatment sequence.
General Health Status and Organ Function
Whether PSMA therapy is appropriate depends not only on the stage of the tumor, but also on the patient’s overall health.
As this is a systemic treatment, certain physical prerequisites must be met. As part of the evaluation of indication, the following factors are considered in particular:
- Bone marrow function
- Kidney function
- General performance status
- Relevant comorbidities
The bone marrow plays an important role, as the therapy can temporarily affect blood cell production. Kidney function is also assessed, since the therapeutic agent is excreted via the kidneys.
These medical requirements are not intended to exclude patients prematurely. Rather, the goal is to use the therapy only when it can be carried out with an acceptable level of risk.
This structured evaluation helps to realistically assess both potential benefits and possible burdens, and to plan treatment responsibly.
A detailed overview of possible therapy-related effects can be found under Side Effects of PSMA Therapy.
Guidelines and Scientific Basis for the Evaluation of Indication
The decision as to whether PSMA therapy is appropriate at Theranosticum Vienna is based on international guidelines and current scientific evidence.
In particular, the recommendations of the European Association of Urology (EAU) include radioligand therapy with Lutetium-177 as a treatment option for advanced, metastatic prostate cancer. This is supported by large clinical studies that have evaluated the effectiveness of the therapy.
For the evaluation of indication, this means:
- The disease must be in an advanced stage
- Previous treatments must be taken into account
- Sufficient PSMA expression must be demonstrated
- The overall health status must allow for treatment
These criteria are not assessed in isolation, but are evaluated within the overall clinical context.
When Is PSMA Therapy Not Appropriate?
Not every patient with advanced prostate cancer meets the requirements for PSMA therapy. A careful evaluation is therefore a central part of the indication assessment.
The therapy is generally not appropriate if:
- No sufficient PSMA expression is detectable on PSMA PET/CT
- Tumor lesions show only low or heterogeneous PSMA uptake
- The disease is still in an early, localized stage
- The general health status does not allow for systemic therapy
- Or if the patient does not consent
Severely impaired kidney or bone marrow function, as well as serious comorbidities, may also make treatment medically inadvisable.
However, a decision against PSMA therapy does not mean that no treatment is possible. In some situations, other therapeutic options may be more appropriate or better tolerated.
Individual Assessment at Theranosticum Vienna
The indication for PSMA therapy at Theranosticum Vienna is determined based on a structured and transparent evaluation of all available findings. Our goal is to realistically assess whether the therapy is appropriate in your individual case.
All relevant information is considered in context, including:
- Current imaging, particularly PSMA PET/CT
- The previous course of treatment
- PSA trends
- Overall health status and existing comorbidities
Our assessment follows established clinical guidelines and incorporates the latest scientific evidence.
If the criteria are met, the therapy can be planned. If the requirements are not fulfilled, this is communicated openly and discussed together. This approach helps avoid unrealistic expectations and supports well-informed decision-making.
Frequently Asked Questions About the Indication for PSMA Therapy
What requirements must be met for PSMA therapy to be appropriate?
PSMA therapy is generally appropriate when advanced or metastatic prostate cancer is present, previous therapies are no longer sufficiently effective, and sufficient PSMA expression has been demonstrated on PSMA PET/CT.
Is PSMA PET/CT a mandatory requirement for the indication?
Yes. PSMA PET/CT is a fundamental requirement. Only if tumor cells express sufficient PSMA can the therapy act in a targeted manner. Without this confirmation, the treatment is generally not appropriate.
Is a rising PSA level alone sufficient for the indication?
No. A PSA increase alone is not sufficient. The decision is based on the overall clinical picture, including imaging, treatment history, clinical condition, and general health status.
Is chemotherapy required before PSMA therapy?
In many cases, PSMA therapy is used after other systemic treatments. Whether prior chemotherapy is required depends on the individual disease course and current clinical guidelines.
When is PSMA therapy not appropriate?
The therapy is generally not appropriate if there is insufficient PSMA expression, the disease is still localized, or the general health status does not allow for systemic treatment.
Who determines the indication for PSMA therapy?
The indication is determined by us following a structured medical evaluation. This assessment takes into account clinical guidelines, current findings, the prior course of therapy, and the overall health status of our patients.
We take time for what matters - your care
If you have questions or would like to schedule an appointment, we are here for you.
In a personal consultation, we discuss your concerns in a calm setting and outline the next steps together.
Our team will get back to you promptly and support you competently and reliably - from the first contact through to your therapy appointment.




